General Information/IS related to Integrative Conjugative Elements (ICEs)
| Other structures which obscure the definition of an IS have been identified among various TEs. For example, ICE (Integrative Conjugative Elements), are found integrated into the host genome but can excise and transfer from cell to cell. Their insertion into and excision from their host chromosomes is generally using a tyrosine-based integrase related to phage integrases[1][2][3] (Fig.7.1) while others possess a serine-recombinase (Fig.7.1 and Fig.11.1).
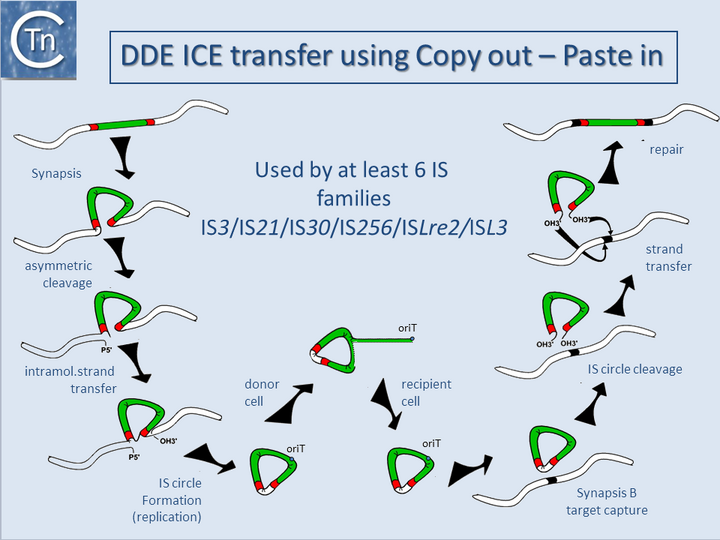
Their transfer depends on a second set of proteins which includes a “relaxase”, often a single strand endonuclease of the HUH superfamily[4][5]. However, ISSag10, a tIS member of the IS1595 family from Streptococcus agalactiae which includes an O-lincosamide nucleotidyltransferase gene, encodes a DDE transposase, nd undergoes cell-to-cell transfer when complemented with an autonomous ICE, Tn916[6]. In this case, a cryptic origin of transfer is located within the 3’ end of the resistance gene. These non-autonomous ICE have been called IME (Integrative Mobilizable Elements[7]) or CIME (CIs-Mobilizable Elements[8]). More recent studies have identified a new ICE family, transposon of Group B Streptococcus (TnGBS), in which the enzyme catalyzing their integration and excision belongs to another DDE-group Tpase [9][10][11][12] (Fig.11.1). This led to the identification of an entirely new family of classic IS carrying DDE Tpases, the ISLre2 family[11]. Other ICE have been identified which include a DDE Tpase closely related to that of the IS30 family[13]]. It is important to note that the IS with these associated DDE transposases transposes using a copy-paste mechanism which involves a double-strand transposon circle. ICE circles are presumably formed using this mechanism in contrast to those possessing tyrosine or serine recombinases. DDE-generated circular ICE would then undergo transfer using the specific relaxase system (Fig.11.2). It seems likely that examples of ICE with other IS family Tpases are awaiting identification. Moreover, in addition to a variety of transfer functions, certain ICE carries plasmid-related replication genes important in ensuring sufficient stability of the transposition intermediates to enable their subsequent integration [10][14]. Early examples of ICE[15] were initially thought to be resistance plasmids and assigned an incompatibility group, incJ. These are maintained as an integrated copy in the host chromosome but can nevertheless give rise to circular copies[16]. This is yet another example of the increasingly indistinct frontiers between phage, plasmids, and transposons[17][18][19] (http://db-mml.sjtu.edu.cn/ICEberg/).
|
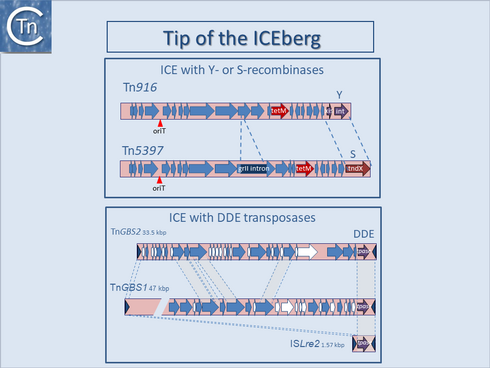 Fig.11.1. Tip of ICEberg. Top panel. The figure shows two closely related ICE that share their conjugation genes, their origins of transfer (red vertical arrowhead), and the tetracycline resistance gene tetM (red arrow) but differs in the type of ‘transposase’ they encode. Tn916 (18032 bp) possesses a bacteriophage lambda-like system including a tyrosine site-specific recombinase (int) and a gene required with int for excision (xis). In Tn5397 (20658 bp) the int/xis system is replaced by a site-specific serine recombinase gene tndX. A group II intron insertion has occurred into one of the conjugation-like genes in Tn5397. Bottom panel. ICE with DDE transposases. The relationship between two ICE from Streptococcus agalactiae and a simple insertion sequence, ISLre2 is shown. |
Bibliography
- ↑ Burrus V, Waldor MK . Shaping bacterial genomes with integrative and conjugative elements. - Res Microbiol: 2004 Jun, 155(5);376-86 [PubMed:15207870] [DOI]
- ↑ Burrus V, Pavlovic G, Decaris B, Guédon G . Conjugative transposons: the tip of the iceberg. - Mol Microbiol: 2002 Nov, 46(3);601-10 [PubMed:12410819] [DOI]
- ↑ Burrus V . Mechanisms of stabilization of integrative and conjugative elements. - Curr Opin Microbiol: 2017 Aug, 38;44-50 [PubMed:28482230] [DOI]
- ↑ Burrus V, Pavlovic G, Decaris B, Guédon G . The ICESt1 element of Streptococcus thermophilus belongs to a large family of integrative and conjugative elements that exchange modules and change their specificity of integration. - Plasmid: 2002 Sep, 48(2);77-97 [PubMed:12383726] [DOI]
- ↑ Chandler M, de la Cruz F, Dyda F, Hickman AB, Moncalian G, Ton-Hoang B . Breaking and joining single-stranded DNA: the HUH endonuclease superfamily. - Nat Rev Microbiol: 2013 Aug, 11(8);525-38 [PubMed:23832240] [DOI]
- ↑ Achard A, Leclercq R . Characterization of a small mobilizable transposon, MTnSag1, in Streptococcus agalactiae. - J Bacteriol: 2007 Jun, 189(11);4328-31 [PubMed:17416666] [DOI]
- ↑ Adams V, Lyras D, Farrow KA, Rood JI . The clostridial mobilisable transposons. - Cell Mol Life Sci: 2002 Dec, 59(12);2033-43 [PubMed:12568329] [DOI]
- ↑ Pavlovic G, Burrus V, Gintz B, Decaris B, Guédon G . Evolution of genomic islands by deletion and tandem accretion by site-specific recombination: ICESt1-related elements from Streptococcus thermophilus. - Microbiology (Reading): 2004 Apr, 150(Pt 4);759-774 [PubMed:15073287] [DOI]
- ↑ Brochet M, Da Cunha V, Couvé E, Rusniok C, Trieu-Cuot P, Glaser P . Atypical association of DDE transposition with conjugation specifies a new family of mobile elements. - Mol Microbiol: 2009 Feb, 71(4);948-59 [PubMed:19183283] [DOI]
- ↑ 10.0 10.1 Guérillot R, Da Cunha V, Sauvage E, Bouchier C, Glaser P . Modular evolution of TnGBSs, a new family of integrative and conjugative elements associating insertion sequence transposition, plasmid replication, and conjugation for their spreading. - J Bacteriol: 2013 May, 195(9);1979-90 [PubMed:23435978] [DOI]
- ↑ 11.0 11.1 Guérillot R, Siguier P, Gourbeyre E, Chandler M, Glaser P . The diversity of prokaryotic DDE transposases of the mutator superfamily, insertion specificity, and association with conjugation machineries. - Genome Biol Evol: 2014 Feb, 6(2);260-72 [PubMed:24418649] [DOI]
- ↑ Dordet Frisoni E, Marenda MS, Sagné E, Nouvel LX, Guérillot R, Glaser P, Blanchard A, Tardy F, Sirand-Pugnet P, Baranowski E, Citti C . ICEA of Mycoplasma agalactiae: a new family of self-transmissible integrative elements that confers conjugative properties to the recipient strain. - Mol Microbiol: 2013 Sep, 89(6);1226-39 [PubMed:23888872] [DOI]
- ↑ Smyth DS, Robinson DA . Integrative and sequence characteristics of a novel genetic element, ICE6013, in Staphylococcus aureus. - J Bacteriol: 2009 Oct, 191(19);5964-75 [PubMed:19648240] [DOI]
- ↑ Lee CA, Babic A, Grossman AD . Autonomous plasmid-like replication of a conjugative transposon. - Mol Microbiol: 2010 Jan, 75(2);268-79 [PubMed:19943900] [DOI]
- ↑ Murphy DB, Pembroke JT . Transfer of the IncJ plasmid R391 to recombination deficient Escherichia coli K12: evidence that R391 behaves as a conjugal transposon. - FEMS Microbiol Lett: 1995 Dec 15, 134(2-3);153-8 [PubMed:8586262] [DOI]
- ↑ Pembroke JT, Murphy DB . Isolation and analysis of a circular form of the IncJ conjugative transposon-like elements, R391 and R997: implications for IncJ incompatibility. - FEMS Microbiol Lett: 2000 Jun 15, 187(2);133-8 [PubMed:10856646] [DOI]
- ↑ Bi D, Xu Z, Harrison EM, Tai C, Wei Y, He X, Jia S, Deng Z, Rajakumar K, Ou HY . ICEberg: a web-based resource for integrative and conjugative elements found in Bacteria. - Nucleic Acids Res: 2012 Jan, 40(Database issue);D621-6 [PubMed:22009673] [DOI]
- ↑ Toussaint A, Merlin C, Monchy S, Benotmane MA, Leplae R, Mergeay M, Springael D . The biphenyl- and 4-chlorobiphenyl-catabolic transposon Tn4371, a member of a new family of genomic islands related to IncP and Ti plasmids. - Appl Environ Microbiol: 2003 Aug, 69(8);4837-45 [PubMed:12902278] [DOI]
- ↑ Faelen M, Toussaint A, Waggoner B, Desmet L, Pato M . Transposition and replication of maxi-Mu derivatives of bacteriophage Mu. - Virology: 1986 Aug, 153(1);70-9 [PubMed:3016987] [DOI]